Last updated on March 5th, 2023 at 05:51 pm
The battery was invented by Alexander Volta in 1800. Although various iterations have happened since then, the fundamental working of a battery is still the same. Batteries provide electrical energy from chemical energy. Thus, the chemical composition inside the battery is very crucial for the perfect functioning of a battery. This article discusses the composition of an alkaline battery and how are they made.
What’s inside a battery?
A battery consists of three major components – the two electrodes and the electrolyte. But the commercial batteries consist of a few more components that make them reliable and easy to use.
In simple words, the battery produces electricity when the two electrodes immersed in the electrolyte react together. Electricity is basically the flow of electrons. The chemical composition of the battery is designed in such a way that the electron from one electrode flows through the electrolyte to the other electrode. This happens only when the battery is connected to the circuit.

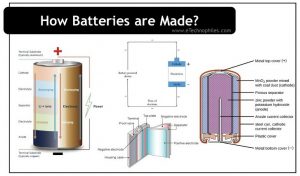
The inner layers of an alkaline battery are shown in the figure above. Let us see the components of a commercial alkaline battery in detail.
Must read: Important Battery Terms & Characteristics Explained (with Examples)
The case
The case is the outermost covering of the battery. It is usually made of thin steel sheets. It acts as a holder and keeps the battery components and insulation away from the ambient. A plastic wrapper is placed over this case which shows us the specifications and the terminals of the battery.
Positive terminal
Note: The positive terminal does not mean the cathode. But generally, both these terms are used interchangeably while discussing battery terminals.
Actually, the cathode is present inside the battery, while the positive terminal of the battery lies outside and is visible to us. The positive terminal connects the cathode to the circuit. In an alkaline battery, the positive terminal is a small projection at one end of the battery.
Negative terminal
Similar to the cathode, the anode also lies inside the battery, while the negative terminal lies outside. The negative terminal connects the anode to the circuit. In an alkaline battery, the negative terminal is the base cap at the other end of the battery. It appears as a flat surface.
Anode
The anode has the capacity to release electrons. Alkaline batteries use zinc as the anode. This metal easily releases electrons. The zinc is mixed with potassium hydroxide solution to form a paste.
Cathode
The cathode accepts the electrons released by the anode. Manganese dioxide is used in alkaline batteries as its cathode. Manganese oxide is mixed with graphite to increase its conductivity.
Electrolyte
The electrolyte is the medium to transfer electrons between anode and cathode. Alkaline batteries use liquid Potassium hydroxide (KOH) as the electrolyte. It is due to this compound that alkaline batteries got their name. KOH is a good conductor and is thus a perfect choice as an electrolyte.
Separator
A separator is an important part of a battery since it saves the battery from an internal short circuit. As its name specifies, a separator keeps the anode and cathode apart. If these electrodes come in direct contact, due to a short circuit the battery will drain out completely. Hence a rubber-like laminated paper is rolled and used as the separator for alkaline batteries.
Current collector nail
The current collector nail is an inevitable part of a battery that finally delivers power from the battery to the gadgets. A brass nail is used as the current collector in the batteries. This nail is placed in the exact middle of the battery and transfers the external current to the internal elements.
Similar Articles on Batteries
The Battery Assembly Process
After discussing the major elements of a battery, let us now see how they are assembled to form a battery that reaches our hands as the final product. Here is the step-by-step process.
Stage 1: Selecting the size of the case
As per the predefined size, the battery container is prepared from nickel-plated steel sheets. For alkaline batteries AA, AAA, AAAA, C, D, etc. are the popular sizes.
Stage 2: Inserting the cathode
As we have already discussed, a mixture of manganese dioxide and graphite is prepared that is used as the cathode. This mixture is compressed into thin pellets or rings. Four rings are filled into each battery can.
Stage 3: Placing the seperator
As soon as the cathode is ready, the next immediate step is to place the separator. It is placed in the middle of cathode rings and is sealed at the bottom.
Stage 4: Filling the liquid electrolyte
Once the separator is fixed, the liquid electrolyte is filled inside. The separator and cathode rings absorb the electrolyte. This process takes about 20 minutes to complete and will be continuously monitored.
Stage 5: Placing the anode
While the electrolyte is absorbed, the anode mixture is prepared. The zinc gel is prepared by mixing it with potassium hydroxide. It takes about 10 minutes to form a light blue paste. Once stage 4 is done, this zinc gel is filled into the separator.
Stage 6: Inserting the current collector
Even if all the elements are kept in place, a battery in this form might not work. For this, a current collector nail is used. It transfers the energy into the battery elements in order to initiate the chemical reaction.
A brass nail is used as the current collector after several rounds of cleaning to ensure it is free from dirt or oil. It is then inserted right inside the battery and is soldered into a steel disc. This steel disc acts as the negative pole of the battery.
Stage 7: Packing the battery
The process is now at the final stage of packing. The finished batteries can now be sealed using high-speed capping. It is then followed by covering the batteries with a plastic wrapper. It includes the specifications and other details of the battery. Once they are packed and transported, they are available for our use.